Real-Time Release Testing of Oral Solid Dosage Forms
ARTIFICIAL INTELLIGENCE FOR DISSOLUTION PREDICTION
NOVEL PAT-BASED RTRT OF DISSOLUTION PERFORMANCE
PROJECT LEAD: Dr. Elisabeth Fink
PROJECT DURATION: December 1, 2021 to November 30, 2024
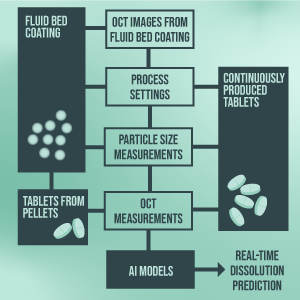
The real-time prediction of the dissolution behavior for solid oral dosage forms is an important area of interest. In the PAT-RTRT project two selected use cases of compressed tablets are considered. First, fluid bed coated pellets are part of a formulation for tablets compressed by Pfizer. Data such as in-line OCT from the coater and other PAT methods and process data, are inputs for a predictive model for the dissolution behavior. In the second stream, RCPE’s own ConsiGma line is used for the production of tablets under different process conditions. Here, the focus is the introduction of novel PAT methods such as Terahertz spectroscopy and GASMAS (gas in scattering media absorption spectroscopy) measurements. Measurements from these tools will be evaluated in their predictive power for dissolution.
Further, a feasibility study will evaluate how in-line ready these technologies are and what steps need to be undertaken to take full advantage of these methods. This will be strongly supported by the University of Cambridge with expert knowledge about Terahertz spectroscopy. As a final ingredient, RCPE’s own OCT solution will be installed for real-time, in-line monitoring of the compressed tablets directly after the tablet press. During modeling, advanced artificial intelligence methods will be used, and predictions will be made based on probabilities for the expected value ranges. The project is completed with regulatory guidelines regarding RTRT.
MAIN OBJECTIVES
01
Evaluation of advanced PAT tools for their feasibility in predicting dissolution data
02
Predictive model for the dissolution of tablets compressed of a formulation including coated pellets
03
Predictive model for the dissolution of compressed tablets with in-line PAT methods from the ConSigma line
Learn more about our other projects:





